Sum of reactants (Δ v rΔHº f(reactants)) = (+454 kJ) + (0. So,Īdd these two values in order to get the sum of the reactants The stoichiometric coefficient of this compound is equal to 5 mole. Please enter either the temperature or the pressure, and click on the Go button to proceed. Multiply this value by the stoichiometric coefficient, which in this case is equal to 2 mole. When one of them is given, the other is determined.
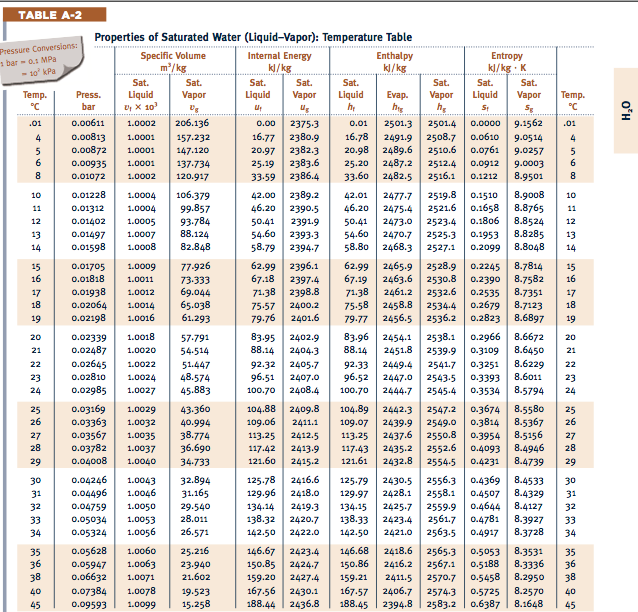
#Water tables thermodynamics calculator how to
What is the specific heat of silver How to use the calculator: Clear the table by pressing the. Now, find the enthalpies of the reactants: The final temperature of the water in the calorimeter is 22.0 C. Sum of products (Σ v pΔHº f(products)) = (-1574 kJ) + (-483.6 kJ) = -2057.6 kJ From the equations derived in this article we can calculate the entropy of wet steam, dry steam and superheated steam. Now add these two values in order to get the sum of the products The stoichiometric coefficient of this compound is equal to 2 mole.
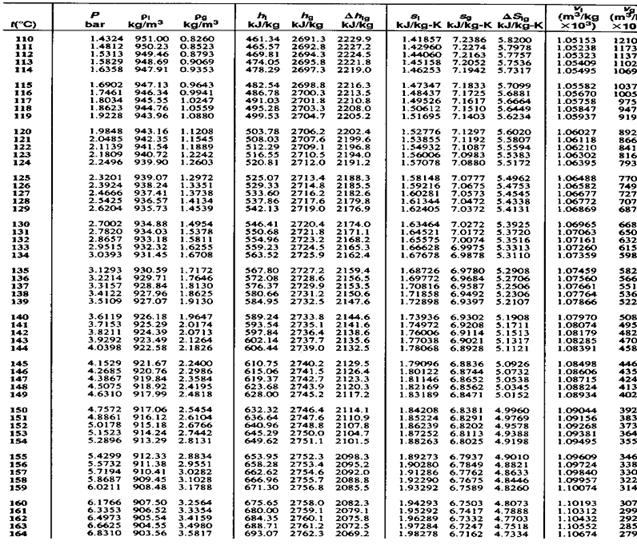
Multiply this value by the stoichiometric coefficient, which in this case is equal to 4 mole. The variable properties for ice, water and steam included in the Steam Tables (where applicable) are enthalpy, entropy, internal energy, density (specific. Because this is above the boiling point of water at 1. \( \newcommand\]Ģ) Next, locate a table of Standard Enthalpies of Formation to look up the values for the components of the reaction (Table 7.2, Petrucci Text)ģ) First find the enthalpies of the products: 03> Calculate the chemical affinity of the formation reactions on an Eh-pH grid at 200C. The data of the site are in good agreement with the National Bureau of Standards (NIST), which contains more than 2000 kinds of substances: water and water vapor, air, refrigerant, hydrocarbon natural gas.
